Our mission is to provide the best thrombectomy system to health care professionals.
Anaconda Biomed S.L. is a Barcelona based medical device start-up co-founded by Dr. Marc Ribo and Dr. Ofir Arad. The company is developing the next generation of catheters to perform safely and efficiently mechanical thrombectomies.
Stroke is the first cause of permanent disability in the world. The burden for the national health systems can be quantified in billions of dollars.
NEXT GENERATION OF CATHETERS
Benefits
A.- FLOW RESTRICTION
Supports the clot attachment to the retrieval devices (stent retrievers) during retrieval:
1.- Reduce the risk of clot detachment and embolization
2.- Increase the efficacy of retrieval devices in mechanical thrombectomy
B.- STRONGEST SUCTION FORCE
1.- Suction (removal/retention force applied over the clot) is a matter of force not of flow
Suction Force vs Flow – Published Paper
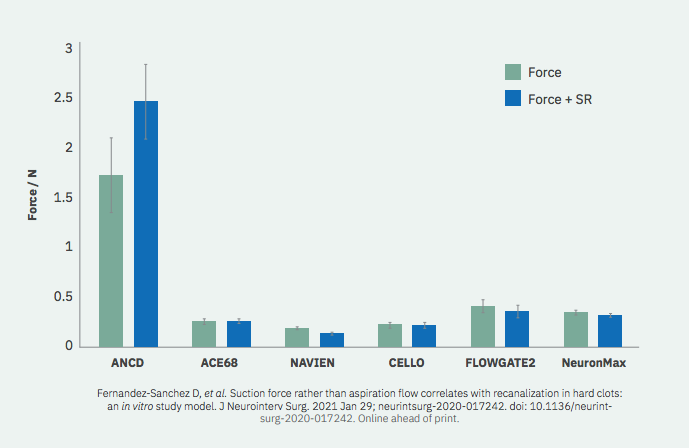
2- Once vacuum is achieved (from 5s on)
3- Depends on radius at 2nd power
4- The bigger the distal end of the catheter, the stronger the force applied over the Clot.
C.- SECURE RETRIEVAL
1.- The nitinol braided funnel will adjust to the artery diameter up to 5mm limiting “shaving” and fragmentation.
2.- The combination of aspiration and stent retrievers is currently the most reliable technic.
3.- Its covered structure will protect it from the flow further reducing potential fragmentation.
D.- SAFE EXITS
1.- Deployed funnel protects the clot during retrieval
2.- Application of aspiration, increases the retention of the clot during the retrieval
E.- RESULTS
A.- IN VITRO STUDIES:
Paper released in JNIS, June 2019. ANCD thrombectomy device: in vitro evaluation
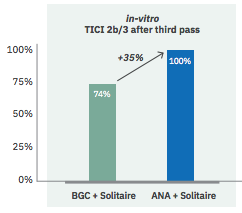
Sanchez S, et al. ANCD thrombectomy device: in vitro evaluation. J Neurointerv Surg. 2020 Jan; 12(1):77-81.
B.- IN VIVO STUDIES:
Paper released in JNIS, January 2020. ANCD thrombectomy device: in vivo evaluation
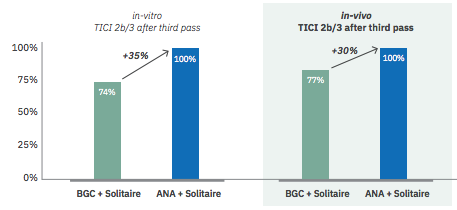
Sanchez S, et al. Preclinical evaluation of the ANCD thrombectomy device: safety and efficacy in a swine clot model. J Neurointerv Surg. 2020 Oct; 12(10):1008-1013
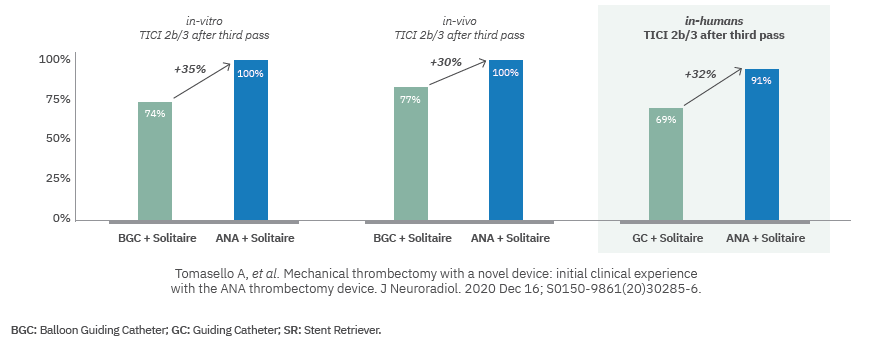
C.- IN HUMAN STUDIES:
Paper released at Journal of Neuroradiology, January 2021. Initial clinical
experience with the ANA thrombectomy device

The ANA is designed for its use in the anterior and posterior neurovascular vessels, such as the Internal Carotid Artery (ICA), the M1 and M2 segments of the middle cerebral artery, the basilar, the posterior cerebral and the vertebral arteries.
Description: The ANA is comprised of two coaxial catheters (Delivery catheter and Funnel catheter) made from variable stiffness sections to be used in conjunction with a Stent Retriever. The funnel catheter is comprised by a radiopaque Nitinol braid covered with a Silicone coating to provide local flow restriction. The delivery catheter has a hydrophilic coating to reduce friction during use and a radiopaque marker on the distal end. Both catheters have a Luer hub on the proximal end. The device is supplied pre-loaded in an insertion tool.

The device is indicated for the introduction of interventional devices into the neuro vasculature, during interventions for restoring blood flow in patients experiencing an acute ischemic stroke due to large vessel occlusion.
Description: It is a sterile, single-use, disposable intravascular device comprised of one catheter (funnel catheter) consisting of sections of variable stiffness. The funnel catheter is comprised of a radiopaque nitinol braid (a self-expanding funnel) covered by a continuous silicone coating that provides local flow restriction when deployed. The catheter has a luer lock hub on its proximal end and it is provided already loaded with the introducer sheath on the catheter ready for its use


